Monel is also used in the frames of eyeglasses and even in motion picture film processing. Monel, an alloy of nickel, copper and small amounts of iron, carbon, manganese, silicon, is preferred in aircraft construction, buildings, oil production, piping systems, musical instruments, the chemical industry. Sterling silver, an alloy of silver and copper, is used in forks, spoons, knives, medical instruments, as well as in the manufacture of saxophones. 
Cuprous sulfide is used in electrodes, solar cells, etc. Cuprous chloride is generally used as a catalyst. Cupric sulfate is mostly used for agricultural purposes. Cuprous oxide is used as a pigment in glasses, porcelain, paints, and ceramics. The melting point of copper is 1084C whereas the boiling point of. Copper and its useful alloys are employed in the medical field and in agriculture. Copper has high density, melting point and boiling point. A The element with 82 protons (atomic number of 82) is lead: Pb. C Give the symbol of each isotope with the mass number as the superscript and the number of protons as the subscript, both written to the left of the symbol of the element. Uses of CopperĬopper, a reddish and ductile metallic element with the symbol Cu, is mainly used as a conductor of electricity and heat. B Calculate the mass number of each isotope by adding together the numbers of protons and neutrons. 
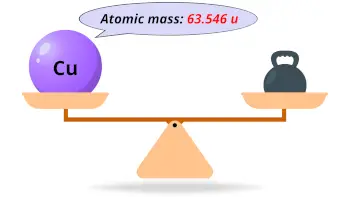
Another famous alloy of copper – the one with zinc – is called brass, and it is widely used for producing parts of musical instruments, screws, buttons, door knobs, and many other things. Together with tin, copper forms an alloy named bronze, which is used for producing kitchenware, valves, gears, electrical components, bells, and so on. ChEBI A heavy metal trace element with the atomic symbol Cu, atomic number 29, and atomic weight 63.55. Besides, copper is extensively used in producing electrical element, especially wires or others. Copper-63 is the stable isotope of copper with relative atomic mass 62.929601, 69.2 atom percent natural abundance and nuclear spin 3/2. Centuries ago, copper was considered to be a precious metal and was used to produce coins, along with gold and silver. 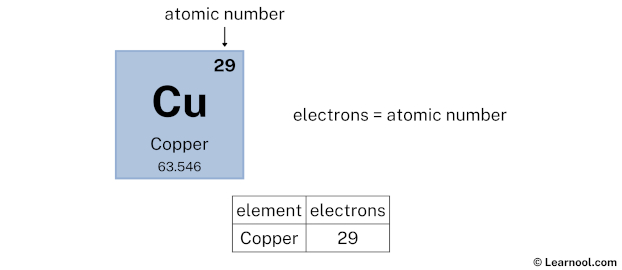
It is possible to find copper in its pure form in nature, but it is usually obtained from natural minerals. Copper is essential for living creatures, and we have it in our bodies in small doses, for proper function of our enzymes. It has got its current name from a Latin word meaning metal from Cyprus. Atomic mass indicated on entries of the Periodic Table. Copper is a reddish transition metal with atomic number 29 and element symbol Cu. The atomic number for copper means that a copper nucleus has 29 protons, and a copper atom has a. On the other hand, the atomic number (Z) of each element is found above the atomic symbol. Copper is atomic number 29 with element symbol Cu. For example, copper has the symbol Cu, and its atomic number, 29, is found above the symbol Cu. The atomic mass of tin (Sn) is 118.71 u while the atomic mass of carbon (C) is 12.011 u. It is known as one of the oldest discovered metals, and humanity worked with copper millions of years ago. The atomic mass of each element is found under the element symbol in the periodic table. Copper is a chemical element of the periodic table with chemical symbol Cu and atomic number 29 with an atomic weight of 63.5463 u and is classed as transition metal and is part of group 11 (coinage metals). To confirm your answer, compare the calculated value to the weighted mass displayed on the periodic table.Copper is a soft metal resembling gold in its color and texture. The atomic mass of oxygen (use percent natural abundance data from Table 2.5.1) would be calculated as follows:Ītomic mass = (%1)(mass 1) + (%2)(mass 2) + (%3)(mass 3) For chlorine, there are only two naturally occurring isotopes so there are only two terms.Ītomic mass = (%1)(mass 1) + (%2)(mass 2) + ⋯Īnother example: oxygen exists as a mixture that is 99.759% 16O, 0.037% 17O and 0.204% 18O. This is repeated until there is a term for each isotope. The weighted average is determined by multiplying the percent of natural abundance by the actual mass of the isotope. The atomic mass of an element is the weighted average of the atomic masses of the naturally occurring isotopes of that element. Copper-65 Cu CID 44150586 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety. Why? The reason is that we need to take into account the natural abundance percentages of each isotope in order to calculate what is called the weighted average. = 35.968 \,u \nonumber \]Īs you can see, the average atomic mass given in the last column of the table above (35.453) is significantly lower. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
